Catalysis
Oxidative/Non-Oxidative Paraffin Dehydrogenation over metal free catalysts
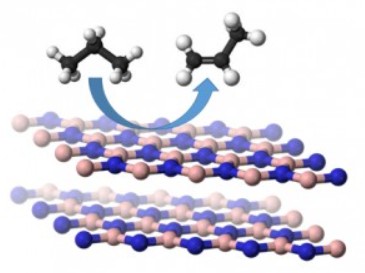
Dehydrogenation of paraffins have been used to synthesize corresponding olefins which are employed as the feedstock for various value added chemicals in polymer, infrastructure, medical and petrochemical industries. In our lab, we have been focusing on novel, metal free, porous layered materials as catalysts with high surface area for dehydrogenation process.
Reference: Piyush C, Momin A and Eswaramoorthy M, Oxidative Dehydrogenation of Propane over a High Surface Area Boron Nitride Catalyst: Exceptional Selectivity for Olefins at High Conversion, ACS Omega, 2018, 3, 369 – 374.
Hydrogen peroxide synthesis
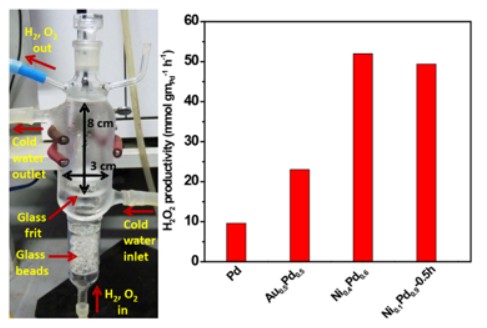
The industrial synthesis of hydrogen peroxide (H2O2) proceeds via an anthraquinone mediated process which is environmentally hazardous. Hence, an alternative way is the direct synthesis of hydrogen peroxide from molecular hydrogen (H2) and oxygen (O2) . It is a challenging reaction in terms of catalyst selection, stability and selectivity. We have been focusing on high surface area, bimetallic systems synthesized by a facile procedure, exhibiting good activity. Reference: Sisir Maity and M. Eswaramoorthy, Nickel-Palladium Bimetallic Catalysts for the Direct Synthesis of H2O2 - Unusual Enhancement of Pd Activity in Presence of Nickel, J. Mater. Chem.A, 2016, 4, 3233 – 3237.
Methane Conversion
Methane is one of the major constituents of natural gas. Conventionally, the conversion of methane to important chemicals proceeds via a two-step energy intensive process. Hence, there is a need to develop an alternative, energy efficient method for the conversion process. The direct conversion of methane to useful chemicals is thus receiving greater attention. In our lab, we are exploring different heterogeneous catalysts based on transition metals which have been reported for efficient methane activation to directly convert methane to methanol with high selectivity.
Formic Acid Decomposition
Formic acid is known as a potential liquid carrier for hydrogen gas. It decomposes via two pathways, one leading to carbon monoxide and water as the product while other providing hydrogen and carbon dioxide. Selective dehydrogenation of formic acid at room temperature, in aqueous medium, in the absence of additives and with minimal noble metal is the need of the hour. In our lab we explore heterogeneous catalyst based systems majorly focusing on amine functionalized materials grafted with metal nanoparticles for selective formic acid dehydrogenation at room temperature. In addition to it, electro catalytic formic acid decomposition is also investigated..
Electrocatalysis
Hydrogen evolution reaction (HER), oxygen evolution reaction (OER) and oxygen reduction reaction (ORR)
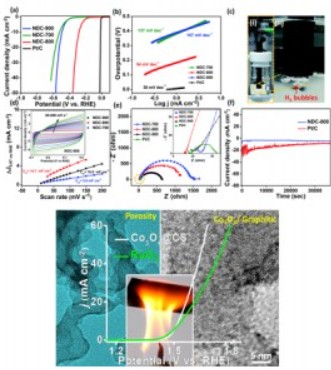
Energy crisis and air pollution are the two major problems that the current human populace is battling with. The need for alternative, cleaner energy sources led to the development of research in fuel cell technology based on H2 economy. Water electrolysis presented itself as an efficient and cleaner source of H2 over the conventional steam reforming process and of O2 as well. OER forms the significant reaction in CO2 reduction (anodic counterpart), metal-air batteries and water splitting. ORR is the cathodic reaction occurring in a fuel cell. However, kinetic barriers endows all of these reactions with the requirement of excess applied electrochemical potential (η, the overpotential). Henceforth, OER, ORR and HER are the electrochemical reactions requiring the development of efficient electrocatalysts operating at lowered overpotential. Electrocatalytic materials based upon carbon, metal oxide-Carbon composites and bimetallic phosphides have already been developed and reported.
References:
1) Dheeraj K S, Soumita C, Arunava S, Sampath S and Eswaramoorthy M, Pick a Wick: A Simple, Ultrafast Combustion Synthesis of Co3O4Dispersed Carbon for Enhanced Oxygen Evolution Kinetics, ACS Applied Energy Materials, 2018, 1, 4448 – 4452.
2) Dheeraj Kumar Singh, RN Jenjeti, S Sampath and M Eswaramoorthy, Two in one: N-doped tubular carbon nanostructure as an efficient metal-free dual electrocatalyst for hydrogen evolution and oxygen reduction reactions, J. Mater. Chem. A, 2017, 5, 6025 – 6031.
Carbon dioxide Reduction
Electrochemical reduction of CO2 serves the dual purpose of reducing CO2 emissions and providing new source of feedstock for industries as well as acting as an alternative fuel source. A huge family of gaseous and liquid products can be obtained ranging from carbon monoxide, acids like formic acid, acetic acid, oxalic acid, alcohols such as ethanol, methanol, hydrocarbons like methane, ethylene to other minor products as well, each having it’s own utility as chemicals and fuels. CO2 electroreduction requires high overpotential necessitating the need for rational maneuvering of electrocatalytic material. Owing to limited solubility of CO2 in aqueous medium which leads to concentration polarization, judicious designing of the electrochemical cell is also of prime importance in this arena. We are currently dealing with investigations related to design and synthesis of bimetallic CO2RR electrocatalysts.
Nitrogen Reduction
In the 20th century, synthesis of ammonia from molecular hydrogen and nitrogen was one of the greatest discovery by Haber and Bosch as ammonia and its derivatives urea is an important component of fertilizer. As the global population is increasing abruptly the need for the fertilizers for food production also increasing day by day. The Haber-Bosch process requires a temperature of 550 deg. C and pressure of more than 200 atm to overcome the sluggish kinetics of the reaction between N2 and H2.. Electrochemical route can be one of the pathway to produce ammonia using the renewable energy sources. But the challenge lies mainly in the selectivity and production rate of ammonia because of competing Hydrogen evolution reaction. In our lab we have optimized the reaction set up for electrochemical nitrogen reduction and procedure for detection of ammonia. We are also exploring various metal (Au,Pd,Ru etc.) and metal oxide based catalysts for better production rate of ammonia and improved faradaic efficiency.
Electrochemical Energy Storage
Hydrogen evolution reaction (HER), oxygen evolution reaction (OER) and oxygen reduction reaction (ORR)
Battery technology is crucial for the future world as a cleaner energy source. A lithium-ion battery is an advanced battery system that uses lithium ions as vital component of its electrochemistry. For electric vehicles and portable electronic applications, the Li-ion secondary batteries have attracted huge attention due to their high electromotive force, high energy density and light weight design. Graphite which is used as an anode material for commercialized lithium-ion battery, cannot fulfill the requirement for high storage capacity due to its insufficient theoretical capacity value of 372 mAh g-1, corresponding to the formation of LiC6 intercalation compound. If the specific capacity is simply increased to 600 mAh g-1 the total battery capacity can exhibit a significant increase in specific capacity by about 1.6 times. Moreover, owing to low abundance of Li metal, attention is being paid towards Na and K-ion batteries. Therefore, our lab is focused onto finding alternate anode materials offering higher discharge capacities like Sn, Ge, Si based materials etc. for Li, Na-ion batteries.
Reference:
1) Subhra G, Dheeraj K Singh, Eswaramoorthy M and Bhattacharya A. J, An Extremely High Surface Area Mesoporous-Microporous-Networked Pillared Carbon for High Stability Li-S and Intermediate Temperature Na-S Batteries, Chemistry Select, 2017, 2, 9249 – 9255.
2) Dheeraj Kumar Singh, SaiKrishna K, S. Harish, S. Sampath and M. Eswaramoorthy, No More HF: Teflon Assisted Ultrafast Removal of Silica to Generate High Surface Area Mesostructured Carbon for Enhanced CO2 Capture and Supercapacitor Performance, Angew. Chem. Int. Ed., 2016, 55, 2032 – 2036.
Drug Delivery
Hydrogen evolution reaction (HER), oxygen evolution reaction (OER) and oxygen reduction reaction (ORR)

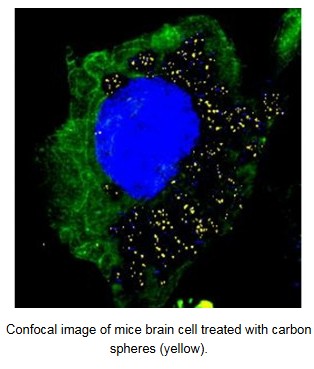
BBB is a highly selective semi-permeable blockade which separates the circulating blood from the brain. So, delivery of drugs in the brain is the major hurdle in the treatment of brain related diseases like Alzheimer’s, Parkinson’s etc. Design of drug delivery materials which can cross BBB is more generic and viable solution compared to drug modification. In our lab, glucose derived carbon spheres were shown to cross BBB and also deliver TTK21 drug molecules (HAT activator) in the brain for the first time. Shape-directed In Vivo compartmentalized delivery of carbon coated iron oxide nanoparticles were also explored.
Reference:
Piyush Chaturbedy, Manoj Kumar, K. Salikolimi, Sadhan Das, H. S. Sarmistha , S. Chatterjee, B. S. Suma, Tapas K Kundu and M. Eswaramoorthy, Shape-Directed Compartmentalized Delivery of a Nanoparticle-Conjugated Small-Molecule Activator of an Epigenetic Enzyme in the Brain , Journal of Controlled Release, 2015, 217, 151 – 159.
Photocatalysis
With increasing urbanisation and industrialisation, the need for energy is also rising. Traditional energy sources like fossil fuels are unreliable because of their carbon footprint and unsustainable nature. Therefore, there is an urgent need for a sustainable green energy source, which leads to chemical energy where energy can be stored in the form of chemical bonds. Hydrogen is one such resource that holds a high amount of energy and is abundant in the Earth's atmosphere as water. It can be obtained by splitting water with an external energy source, which can be in the form of heat, light, pressure, or electricity. In photocatalysis, we utilise abundant energy sources like sunlight to drive reactions that split water molecules to produce hydrogen, which can then be utilised further. Our laboratory also focuses on developing materials that are capable of carrying out photocatalysis.
Aminoclay
Aminoclay and fly ash are promising materials for controlled-release fertilizers (CRFs) due to their unique properties. Aminoclay, with its high surface area and nutrient-binding capacity, is used to form urea nanocomposites, while fly ash, rich in silica and alumina, is used to make cost-effective urea pellets. In our lab, we are independently evaluating the controlled-release performance of these two formulations to assess their potential in reducing nutrient loss and promoting sustainable agriculture.
